QT and QTc intervals
The QT interval is a measure of ventricular depolarization and repolarization on the surface electrocardiogram (ECG), extending from the onset of the QRS complex to the end of the T wave. It reflects the duration of the ventricular action potential and is a key biomarker of cardiac repolarization. Prolongation of the QT interval is clinically important because it is associated with an increased risk of polymorphic ventricular tachycardia, particularly torsade de pointes, which may result in syncope, ventricular fibrillation, or sudden cardiac death. For this reason, evaluation of QT interval prolongation is a central component of the cardiac safety assessment for non-antiarrhythmic drugs.
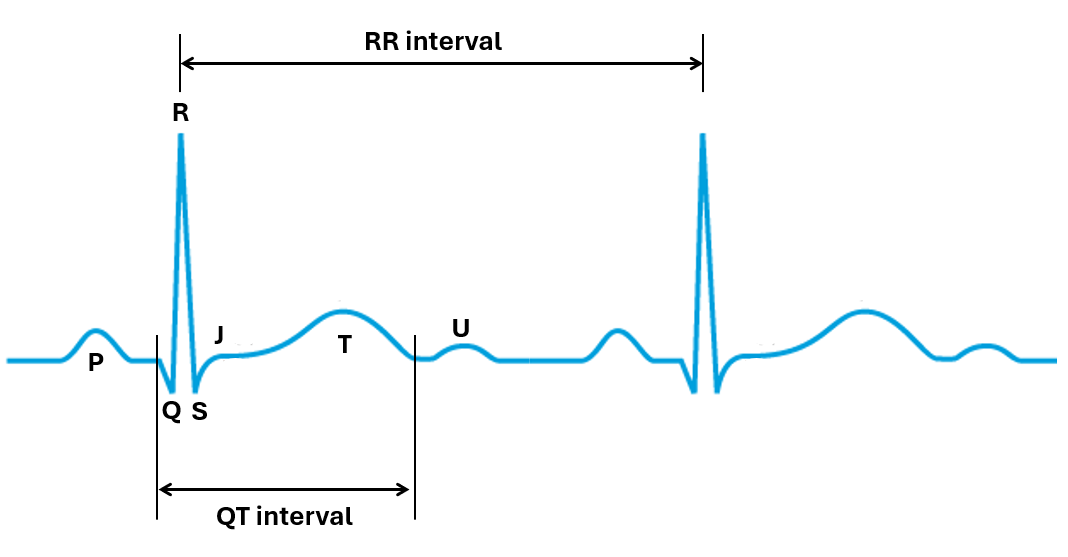
The QT interval is intrinsically dependent on heart rate, which is quantified by the RR interval, defined as the time between two consecutive R waves on the ECG. As heart rate increases (shorter RR interval), the QT interval shortens, whereas at lower heart rates (longer RR interval), the QT interval lengthens. This physiological relationship between QT and RR intervals complicates direct comparison of raw QT measurements across different time points and treatment conditions. To account for this dependency, the QT interval is corrected for heart rate using the RR interval, resulting in the corrected QT (QTc) interval.
Proarrhythmic risk assessment
Historically, drug-induced QTc prolongation was primarily evaluated in dedicated thorough QT (TQT) studies, as described in the International Council for Harmonisation (ICH) E14 guideline Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs. While TQT studies are designed to provide high sensitivity through the use of placebo and positive controls, they typically require large sample sizes and a dedicated study conducted late in development.
Subsequent ICH E14 Q&A documents and regulatory experience have clarified that conc-QTc analysis using high quality ECG measurements of early phase 1 clinical studies can provide an equally robust assessment of QTc liability when appropriately implemented. Regulatory agencies, including the FDA, have explicitly endorsed conc-QTc analysis as a primary strategy for QTc evaluation.
conc-QTc methodology
The most widely applied conc-QTc methodology is based on the linear mixed-effects model described in the IQ Consortium–Cardiac Safety Research Consortium (IQ-CSRC) white paper by Garnett et al. This standard model incorporates baseline QTc, time effects, treatment effect and drug concentration as fixed effects, with random effects to account for inter-individual variability. The slope of the concentration–QTc relationship (drug effect) is the primary parameter of interest and is used to predict the mean QTc effect and its associated two-sided 90% confidence interval at prespecified concentration levels. A lack of clinically meaningful QTc prolongation is typically concluded when the upper bound of this confidence interval remains below the regulatory threshold of concern, generally 10 ms, at the concentrations of interest.

